FOREWORD
“The structure of the helium atom, the existence of half-quantum numbers in band spectra, the continuous spatial distribution of photo-electrons, and the phenomenon of radioactive disintegration, to mention only a few examples, are achievements of the new theory which had baffled the old.”
“This symmetry of the book with respect to the words <particle> and <wave> shows that nothing is gained by discussing fundamental problems (such as causality) in terms of one rather than the other.”
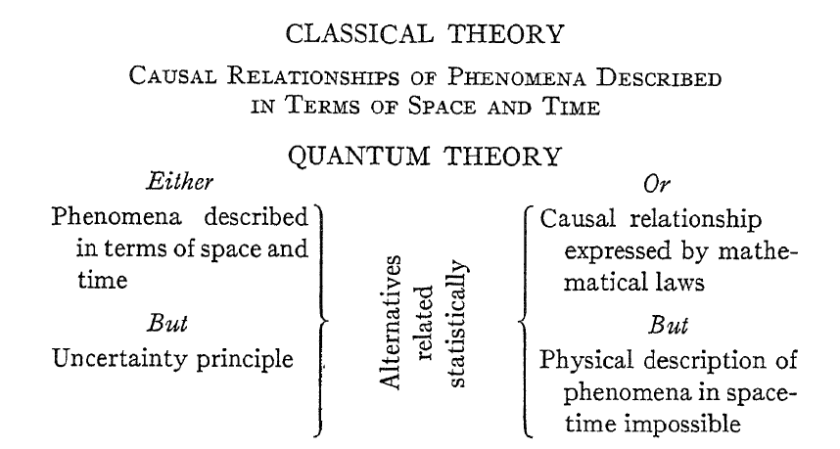
1. INTRODUCTION
“Thus it was characteristic of the special theory of relativity that the concepts <measuring rod> and <clock> were subject to searching criticism in the light of experiment (…) A much more radical departure from the classical conception of the world was brought about by the general theory of relativity, in which only the concept of coincidence in space-time was accepted uncritically. According to this theory, classical concepts are applicable only to the description of experiments in which both the gravitational constant and the reciprocal of the velocity of light may be regarded as negligibly small.
Although the theory of relativity makes the greatest of demands on the ability for abstract thought, still it fulfills the traditional requirements of science in so far as it permits a division of the world into subject and object (observer and observed) and hence a clear formulation of the law of causality. This is the very point at which the difficulties of the quantum theory begin. In atomic physics, the concepts <clock> and <measuring rod> (comprimento) [ou seja, tempo e espaço] need no immediate consideration, for there is a large field of phenomena in which i/c is negligible. The concepts <space-time coincidence> and <observation>, on the other hand, do require a thorough revision.” “the interaction between observer and object causes uncontrollable and large changes in the system being observed, because of the discontinuous changes characteristic of atomic processes.” “it appears that in many cases it is impossible to obtain an exact determination of the simultaneous values of 2 variables, but rather that there is a lower limit to the accuracy with which they can be known.” “These uncertainty relations give us that measure of freedom from the limitations of classical concepts which is necessary for a consistent description of atomic processes.”
“After Newton’s objections to the wave theory of light had been refuted and the phenomena of interference explained by Fresnel, this theory dominated all others for many years, until Einstein pointed out that the experiments of Lenard on the photo–electric effect could only be explained by a corpuscular theory.”
“When the energy of the atom is known, one speaks of a <stationary state of the atom>. When the kinetic energy of the electron is too small to change the atom from its stationary state to a higher one, the electron makes only elastic collisions with the atoms, but when the kinetic energy suffers for excitation some electrons will transfer their energy to the atom, so the electronic current as a function of the velocity changes rapidly in the critical region. The concept of stationary states, which is suggested by these experiments, is the most direct expression of the discontinuity in all atomic processes.”
“It is true that it might be postulated that 2 separate entities, one having all the properties of a particle, and the other all the properties of wave motion, were combined in some way to form <light>. But such theories are unable to bring about the intimate relation between the two entities which seems required by the experimental evidence.” “The solution of the difficulty is that the two mental pictures which experiments lead us to form – the one of particles, the other of waves – are both incomplete and have only the validity of analogies which are accurate only in limiting cases.” “Light and matter are both single entities, and the apparent duality arises in the limitations of our language.” “Fortunately, mathematics is not subject to this limitation” “In order to avoid obscuring the essential relationships by too much mathematics, however, it has seemed advisable to relegate this formalism to the Appendix [que eu, claro, não cubro aqui].”
2. CRITIQUE OF THE PHYSICAL CONCEPTS OF THE CORPUSCULAR THEORY
“As Bohr has emphasized, if a measurement of its co-ordinate is to be possible at all, the electron must be practically free.” “This may be expressed in concise general terms by saying that every experiment destroys some of the knowledge of the system which was obtained by previous experiments.” “It is a matter of personal belief whether such a calculation concerning the past history of the electron can be ascribed any physical reality or not.”
O RABO É MAIS VELOZ QUE A CABEÇA DA COBRA, E AO MESMO TEMPO NÃO É: “The orbit is the temporal sequence of the points in space at which the electron is observed. As the dimensions of the atom in its lowest state are of the order 10-8 cm, it will be necessary to use light of wave-lenght not greater than 10-9 cm in order to carry out a position measurement of sufficient accuracy for the purpose. A single photon of such light is, however, sufficient to remove the electron from the atom, because of the Compton recoil. Only a single point of the hypothetical orbit is thus observable. One can, however, repeat this single observation on a large number of atoms, and thus obtain a probability distribution of the electron in the atom.” “This result is stranger than it seems at first glance. (…) there is thus always a small but finite probability of finding the electron at a great distance from the center of the atom.” “This paradox also serves as a warning against carrying out the <statistical interpretation> of quantum mechanics too schematically. Because of the exponential behavior of the Schrödinger function at infinity, the electron will sometimes be found as much as, say, 1cm from the nucleus. One might suppose that it would be possible to verify the presence of the electron at such a point by the use of red light [faixa de onda mais extensa]. This red light would not produce any appreciable Compton recoil and the foregoing paradox would arise once more.” “The statistical predictions of quantum theory are thus significant only when combined with experiments which are actually capable of observing the phenomena treated by the statistics.”
3. CRITIQUE OF THE PHYSICAL CONCEPTS OF THE WAVE THEORY
“historically we first encounter attempts to develop 3D wave theories that could be readily visualized (Maxwell and de Broglie waves). (…) The reader must be warned against an unwarrantable confusion of classical wave theory with the Schrödinger theory of waves in a phase space.”
“Although it is perhaps possible in principle to diminish these space and time intervals without limit by refinement of the measuring instruments, nevertheless for the physical discussion of the concepts of the wave theory it is advantageous to introduce finite values for the space and time intervals involved in the measurements and only pass to the limit zero for these intervals at the end of the calculations. (…) It is possible that future developments of the quantum theory will show that the limit zero for such intervals is an abstraction without physical meaning” Voilà!
4. THE STATISTICAL INTERPRETATION OF QUANTUM THEORY
“It is instructive to compare the mathematical apparatus of quantum theory with that of the theory of relativity. In both cases there is an application of the theory of linear algebras. One can therefore compare the matrices of quantum theory with the symmetric tensors of the special theory of relativity. The greatest difference is the fact that the tensors of quantum theory are in a space of infinitely many dimensions, and that this space is not real but imaginary.” “The exact knowledge of the numerical value of any dynamical variable corresponds to the determination of a definite direction in this space, in the same manner as the exact knowledge of the moment of inertia of a solid body determines the principal direction to which this moment belongs (it is assumed that there is zero degeneracy).”
“An atom in a (non-degenerate) stationary state presents such a determinate case: The direction characterizing it is that of the kth principal axis of the tensor E, which belongs to the energy value Ekk.” “The total angular momentum of the atom, e.g., can be determined simultaneously with its energy.”
“It follows from this discussion that the value of q’ cannot be uniquely predicted from the result of the experiment determining E, for a disturbance of the system, which is necessarily indeterminate to a certain degree, must occur between the 2 experiments involved. This disturbance is qualitatively determined, however, as soon as one knows that the result is to be an exact value of q. In this case, the probability of finding a value q’ after E has been measured is given by the square of the cosine of the angle between the original direction Ek and the direction q’. (…) This assumption is one of the formal postulates of quantum theory and cannot be derived from any other considerations. It follows from this axiom that the values of 2 dynamical quantities are casually related if, and only if, the tensors corresponding to them have parallel principal axes.”
“Thus one becomes entangled in contradictions if one speaks of the probable position of the electron without considering the experiment used to determine it (cf. the paradox of negative kinetic energy, chapter 2).”
“If one were to treat the measuring device as a part of the system – which would necessitate an extension of the Hilbert space – then the changes considered above as indeterminate would appear determinate. (…) For these observations, however, the same considerations are valid as those given above [um sistema que contém outro sistema enviesa o experimento], and we should be forced, e.g., to include our own eyes as part of the system, and so on. The chain of cause and effect could be quantitatively verified only if the whole universe were considered as a single system – but then physics has vanished, and only a mathematical scheme remains. The partition of the world into observing and observed system prevents a sharp formulation of the law of cause and effect. (The observing system need not always be a human being; it may also be an inanimate apparatus, such as a photographic plate.)”
“With the advent of Einstein’s relativity theory it was necessary for the 1st time to recognize that the physical world differed from the ideal world conceived in terms of everyday experience. It became apparent that ordinary concepts could only be applied to processes in which the velocity of light could be regarded as practically infinite. The experimental material resulting from modern refinements in experimental technique necessitated the revision of old ideas and the acquirement of new ones, but as the mind is always slow to adjust itself to an extended range of experience and concepts, the relativity theory seemed at first repellantly abstract. Nonetheless, the simplicity of its solution of a vexatious problem has gained it universal acceptance. As is clear from what has been said, the resolution of the paradoxes of atomic physics can be accomplished only by further renunciation of old and cherished ideas. Most important of these is the idea that natural phenomena obey exact laws – the principle of causality. In fact, our ordinary description of nature, and the idea of exact laws, rests on the assumption that it is possible to observe the phenomena without appreciably influencing them. To co-ordinate a definite cause to a definite effect has sense only when both can be observed without introducing a foreign element disturbing their interrelation. The law of causality, because of its very nature, can only be defined for isolated systems, and in atomic physics even approximately isolated systems cannot be observed. (…) There exist no infinitesimals by the aid of which an observation might be made without appreciable perturbation.”
“Faraday and Maxwell explained electromagnetic phenomena as the stresses and strains of an ether, but with the advent of the relativity theory, this ether was dematerialized; the electromagnetic field could still be represented as a set of vectors in space-time, however. Thermodynamics is an even better example of a theory whose variables cannot be given a simple geometric interpretation.”
Os matemáticos apertam a mão de Sócrates: nasce a Estatística Aplicada, vulgo cobertor curto da realidade!
“Many of the abstractions that are characteristic of modern theoretical physics are to be found discussed in the philosophy of past centuries. At that time these abstractions could be disregarded as mere mental exercises by those scientists whose only concern was with reality, but today we are compelled by the refinements of experimental art to consider them seriously.”
5. DISCUSSION OF IMPORTANT EXPERIMENTS
“It is true that an ingenious combination of arguments based on the correspondence principle can make the quantum theory of matter together with a classical theory of radiation furnish quantitative values for the transition probabilities, i.e., either by the use of Schrödinger’s virtual charge density or its equivalent, the element of the matrix representing the electric dipole moment of the atom. Such a formulation of the radiation problem is far from satisfactory and leads to false conclusions.”
“Dirac, in his radiation theory, employs the language of the particle representation, but makes use of conclusions drawn from the wave theory of radiation in his derivation of the Hamiltonian function.”
“the classical wave theory is sufficient for the discussion of all questions of coherence and interference.”
“It is very difficult for us to conceive the fact that the theory of photons does not conflict with the requirements of the Maxwell equations. (…) whenever an experiment is capable of furnishing information regarding the direction of emission of a photon, its results are precisely those which would be predicted from a solution of the Maxewell equations of the needle type (unidirectional beams)”
“If one supposes that an experiment has determined the position of the atom with a given accuracy (the value of the momentum must then be correspondingly uncertain), then this means that the density is given by the foregoing expression only in a finite volume v, and is zero elsewhere.” “This example illustrates very clearly how the quantum theory strips even the light waves of the primitive reality which is ascribed to them by the classical theory.”
“For a point electron (one of zero radius) even the classical theory yields an infinite value of the energy, as is well known, so that it becomes necessary to introduce a universal constant of the dimension of a lenght – the <radius of the electron>. It is remarkable that in the non-relativistic theory this difficulty can be avoided in another way – by a suitable choice of the order of non-commutative factors in the Hamiltonian function. This has hitherto not been possible in the relativistic quantum theory.
The hope is often expressed that after these problems have been solved the quantum theory will be seen to be based, in a large measure at least, on classical concepts. But even a superficial survey of the trend of the evolution of physics in the past 30 years [Hallo, século XX!] shows that it is far more likely that the solution will result in further limitations on the applicability of classical concepts than that it will result in a removal of those already discovered.” thenDANsING d SONG O’ THE ‘SIR. ENS’! S.O.[CRATE’]S. FLU-TE & HARP BE4 THE HEM–LOCK… LOCK ‘EM ALL, THE TRUTHS, WITH STRINGS!

